- The SA Health Products Regulatory Authority has cautioned against the use of contaminated children's medication.
- Four products manufactured by an Indian pharmaceutical company have been linked to the deaths of 33 children in the Gambia.
- The World Health Organisation says the company has not assured it of the safety of its products.
The SA Health Products Regulatory Authority (Sahpra) has cautioned against contaminated baby products linked to the deaths of 33 babies.
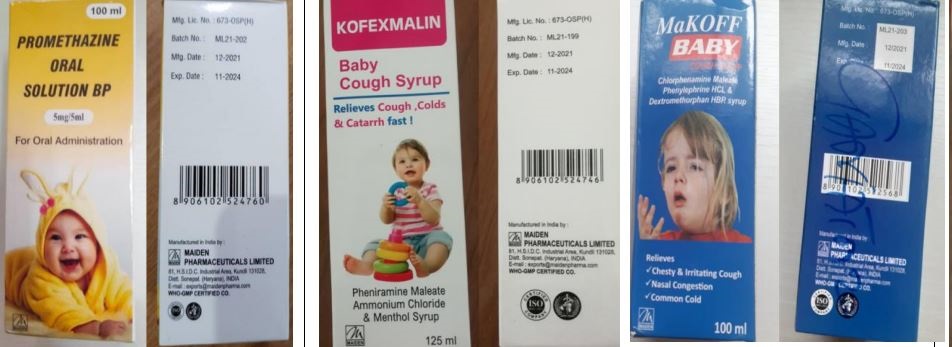
Promethazine Oral Solution, Kofexmalin Baby Cough Syrup, Makoff Baby Cough Syrup, and Magrip N Cold Syrup are products of an Indian company, Maiden Pharmaceuticals.
Sahpra said the World Health Organisation (WHO) had detected the products in the Gambia.
"Thirty-three children in the Gambia died of kidney-related illness. The WHO conducted laboratory tests that linked the medication to those deaths. The four products contain unacceptable amounts of diethylene glycol and ethylene glycol, which are toxic substances and can prove fatal," said the regulator.
READ | Some baby powder products recalled in SA as asbestos is detected
It added side-effects linked to the products, include abdominal pain, vomiting, diarrhoea, inability to pass urine, headache, altered mental state, and acute kidney injury.
Sahpra CEO Dr Boitumelo Semete-Makokotlela said the regulator would work with law enforcement to remove the products.
The WHO added the products did not meet quality standards and the pharmaceutical company had failed to assure it of their safety.
It cautioned the products might have been distributed to other countries illegally.
"If you have these substandard products, please do not use them. If you, or someone you know, have used these products, or suffered any adverse reaction or event after use, you are advised to seek immediate medical advice from a qualified healthcare professional and report the incident to the national regulatory authority," said the WHO.




 Publications
Publications
 Partners
Partners























